The Portuguese pharmaceutical authority, Infarmed, has announced the immediate suspension of the sale of certain batches of the drug Sertraline Generis 50 mg and 100 mg. The decision follows the detection of an impurity exceeding acceptable limits.
“Infarmed is ordering the immediate halt of sales of these batches,” declared in a statement the reference to “Sertraline Generis 50 mg and 100 mg” medications, manufactured by Generis Farmacêutica.
Infarmed emphasized that “entities in possession of these medication batches must not sell, dispense, or administer them and should arrange for their return.” However, patients currently taking these medications “should not stop their treatment” but “should contact their healthcare provider as soon as possible” for an assessment regarding the necessity of switching to an alternative medication.
The affected batches are listed below:
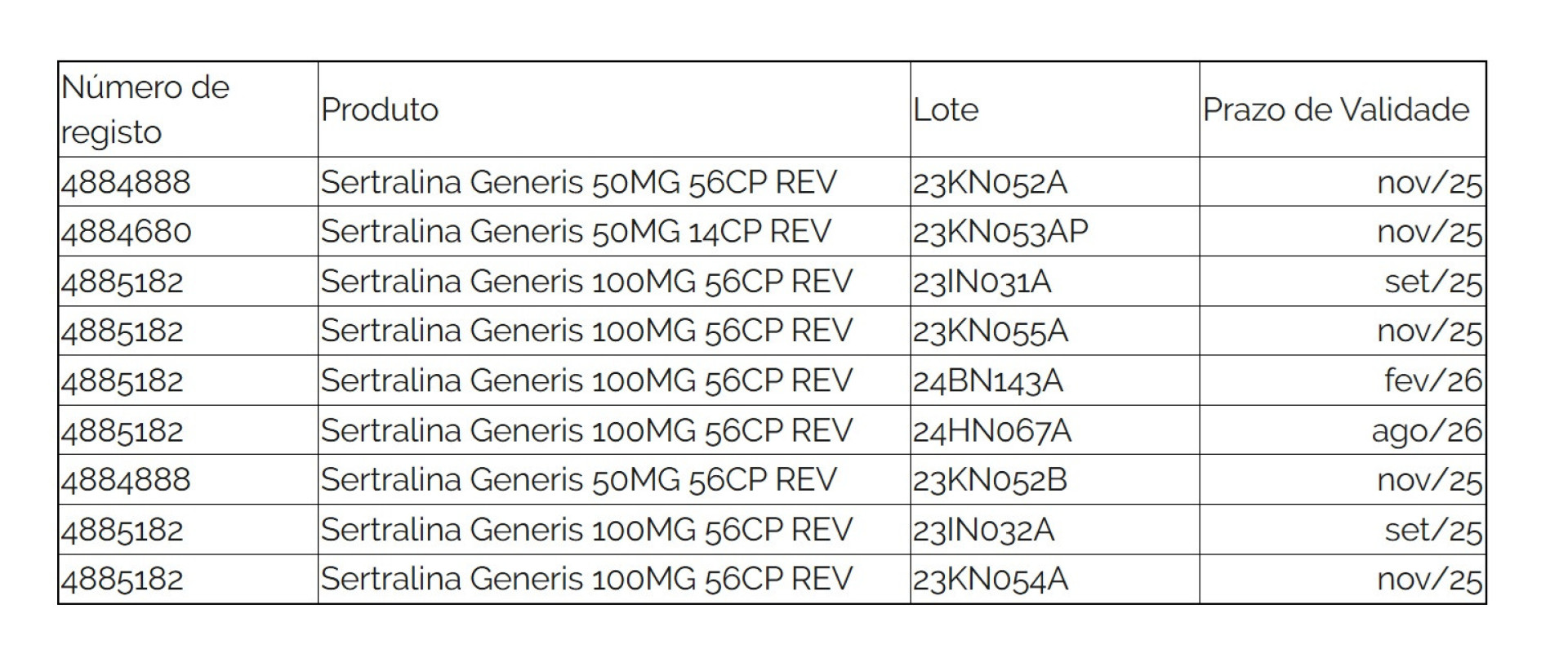
© Infarmed
The medication in question, which is film-coated, was found to have an “impurity exceeding the acceptable limit.”
Sertraline belongs to a class of drugs known as Selective Serotonin Reuptake Inhibitors (SSRIs) and is commonly used to treat depression and/or anxiety disorders.



